The AI-native DCT platform for
behavioral health trials
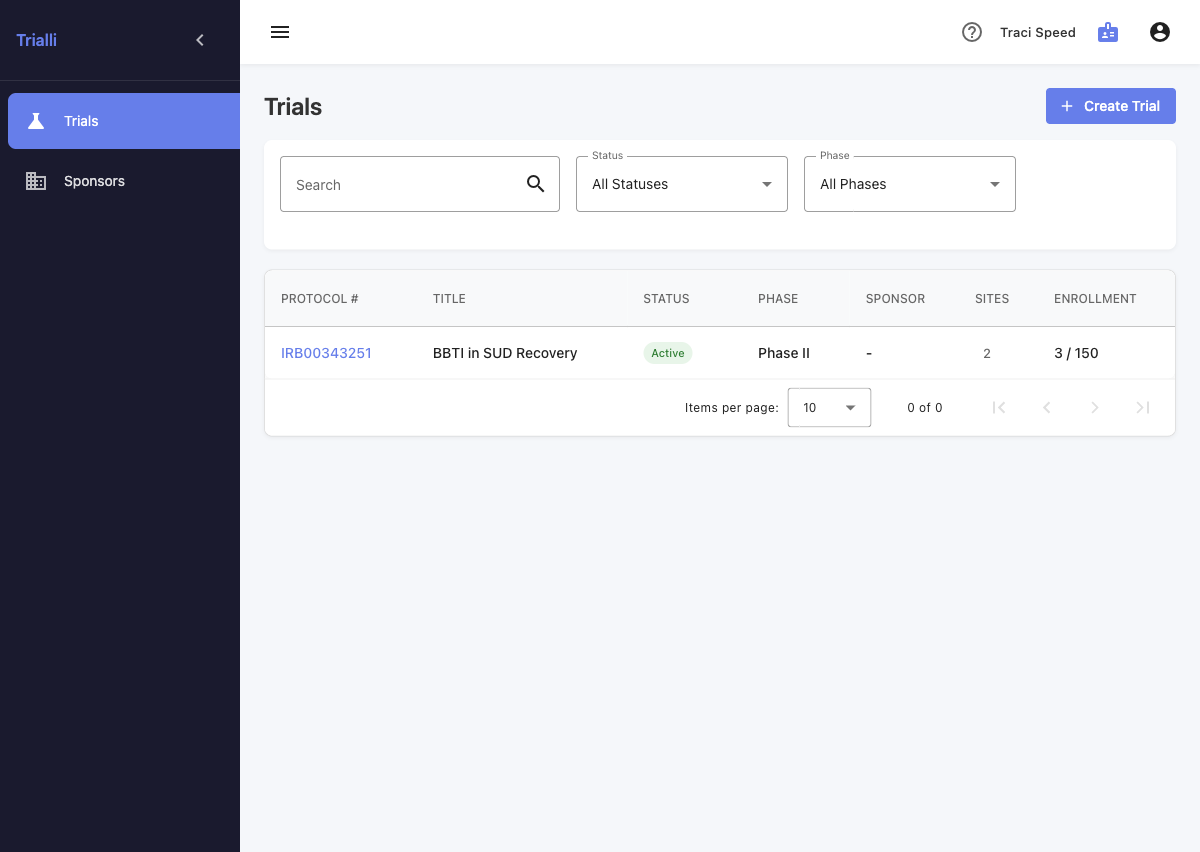
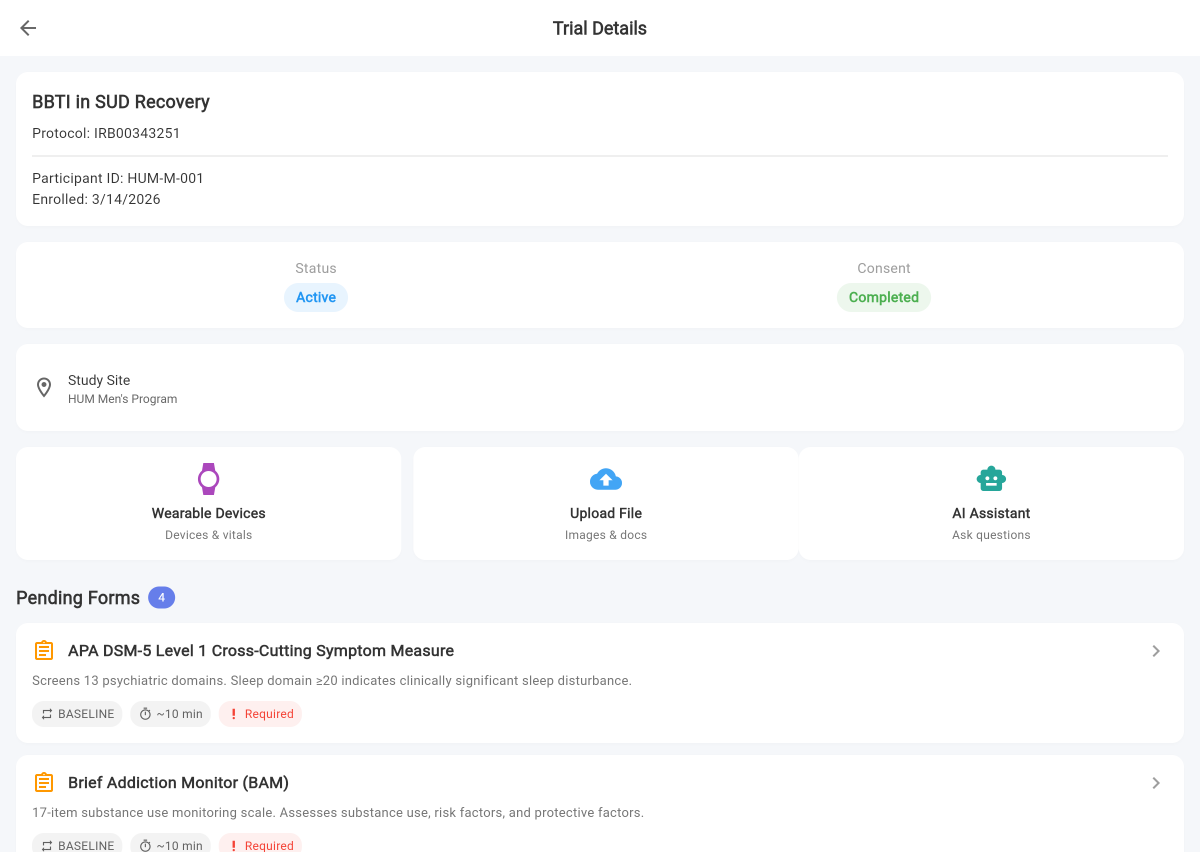
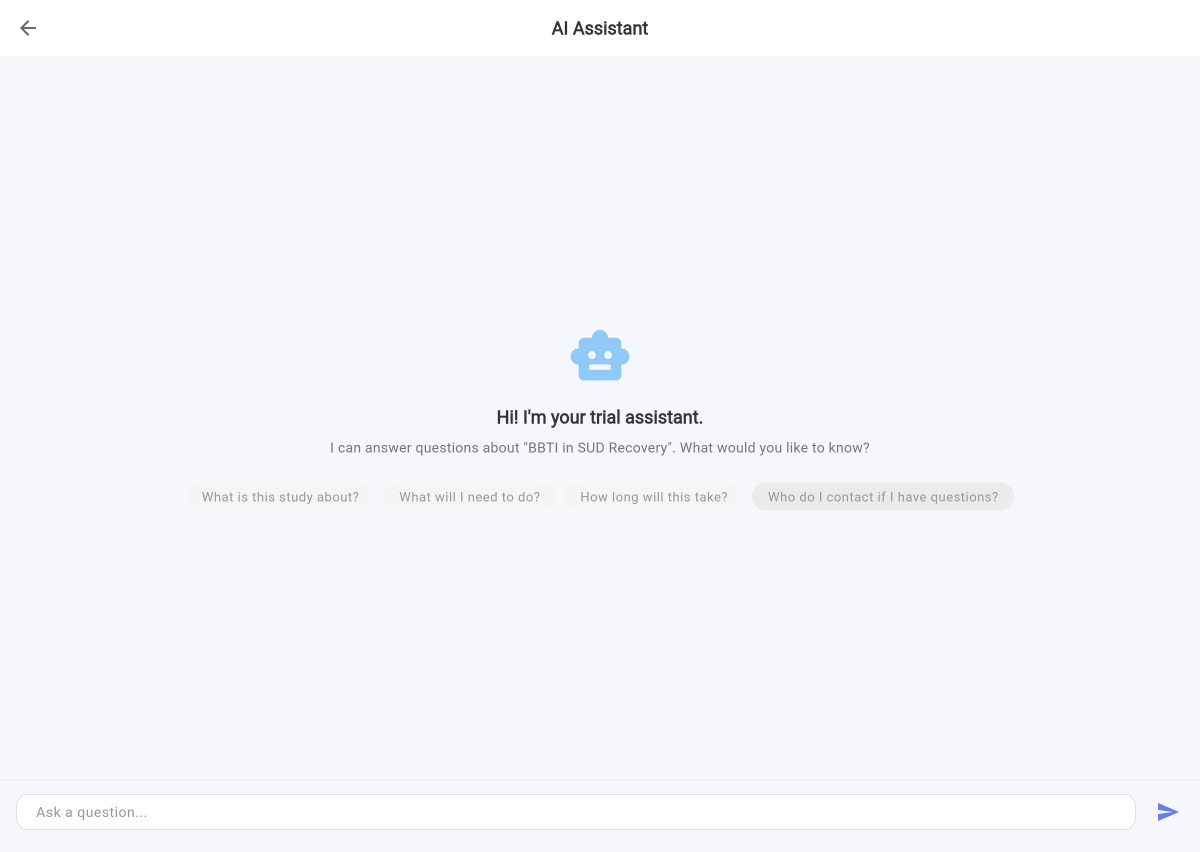
Trialli is the first decentralized clinical trial platform purpose-built for substance use disorder research. AI imports your protocol, captures participant responses by voice, and runs your SUD trial — so coordinators focus on science instead of transcription.
competitors
by 2030
dropout rate
funded